Olefins Primer Series 1: Intro
15-page dive into the heart of the Chemicals industry

The Chemical industry is undergoing its longest and most vicious downcycle in recent history. Margins and utilization have rarely been so low for so long, and company prices reflect this.
At the center of this downcycle is the olefins and polyolefins industry, the largest chemical segment by far, and the heart of the petrochemicals and plastics industry. Many large chemical companies have olefin and polyolefin operations, and their economics make or break downstream industries.
In this Primer series, I want to analyze this industry in order to find opportunities among the chaos, while avoiding the value traps.
The first article of this series serves as a fairly complete introduction to the industry and will serve as a tool to analyze assets and companies in future deliveries. I explain what olefins and polyolefins are, their role in modern society, where and why they are demanded, where they are produced, how their economics work, and why it is undergoing a vicious downcycle.
I also analyze the main trends shaping the cycle of the future: feedstock and competition drivers, the role of climate change and O&G majors, geopolitical uncertainties, and more.
I am certain that after reading this article, you will have a good generalist understanding of where the industry is and where it might head, and that you will have the tools to analyze names and opportunities.
Without further ado, let’s crack it!
Subscribe, it’s free!
Understanding the basics
Olefins are the most important feedstocks into the petrochemical industry. Ethylene and propylene are the building blocks of petrochem (there are other olefins like butene, pentene, etc. with more limited applications).
Polyolefins (polyethylene and polypropylene), the direct derivatives of olefins make up 50% of all plastics globally. Olefins are also present in the production processes of all other important plastics (PS, PVC, PET, PUR).
As building blocks, olefins also mark the limit between the petrochemical industry and the refining industry. Upstream products (like ethane, propane, or naphtha) are considered part of refining. Olefins and their downstream are part of the petrochemical industry.
In this section, we will go over the basics needed to understand the olefins and polyolefins market: what they are, what they are used for, how and where they are manufactured. This section is very important to understand the dynamics of the market later, and to determine company-specific strategic-fit.
Demand sources: the middle class commodity
Olefin demand is tied to plastics consumables and grows very linearly with GDP
Olefins are basic building blocks for all plastics, but two plastics in particular dominate their demand. Polyethylene makes up 60/70% of all demand for ethylene, and polypropylene makes up the same amount of all demand for propylene.
Most polyethylene demand comes from packaging or films for consumer products (the majority of food packaging and plastic bags are made out of polyethylene), plus some piping (construction). Ethylene is also important in the chains of PET (plastic bottles, polyester fabrics) and PVC (pipes, i.e. construction).
Polypropylene is present in many hard plastics products, like a plastic chair, a bottle cap, or a plastic bucket, or as hard pieces in cars or other machinery. Propylene is also part of the chain of ABS (hard plastics), and polyurethanes (construction materials, foams).
That is, the majority of downstream demand for olefins comes from consumables and construction. Urbanization, in particular, is a huge driver of plastics consumption because of the requirements on food and product logistics, construction activity, and transportation, all of which require plastics.
Further, wealth drives plastics consumption linearly. The OECD found that plastics consumption to GDP is almost flat across different countries, at about 1 million tons per $3.5 million in PPP GDP (probably higher now as the figure comes from 2019). Depending on the source, the different plastics types are said to grow between 1.2x and 1.5x GDP, faster in developing countries, and more slowly in developed countries.
Final demand in the rich world, intermediate demand in Asia
The above also means that plastics final consumption centers are the GDP centers: North America, the European Union, developed Asia and China as big drivers, with the emerging populated markets (India, SEA, Africa, LatAm) as potential future growth drivers.
Of course, the fact that these plastics are consumed based on GDP does not mean they are produced where they are consumed, or that their original demand is there. For example, even though a chair made of polypropylene is ‘consumed’ in the US, it may have been made in China, and therefore the polypropylene was ‘consumed’ there. From this perspective, China alone makes up 1/3 of polypropylene intermediate demand and 1/3 to 1/2 of polyethylene intermediate demand, with no European country in the top 10, and the US at only about 10% and 5% of each plastic. Plastic intermediate demand centers skew significantly more Asian along with industrial production.
If you like this, share it, it’s free!
Supply side
Chemistry basics
Chemistry and the upstream petrochemical industry play a relevant role in olefin economics and supply. We need a basic knowledge of feedstocks, logistics, and the chemistry of it all.
Olefins and polyolefins are petrochem derivatives. The chain starts with feedstocks (ethane, propane, naphtha), which are basic chains of carbon and hydrogen. These feedstocks are produced in conjunction with oil and natural gas extraction and refining, as co-products.
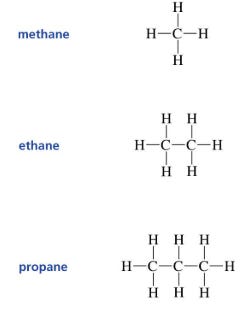
These feedstocks are then cracked in an olefin cracker. They are mixed with steam and heat at super high speeds. This process breaks the molecules, leaving carbon atoms with less hydrogens than needed for stabilization. The carbon atoms join again, but this time with a double (weaker) bond (see image below). The stream is quickly cooled down (quenching) and separated in distillation towers (the classical elongated and consecutive towers of a refining or olefins plant). The results are olefins (ethylene, propylene, butenes).
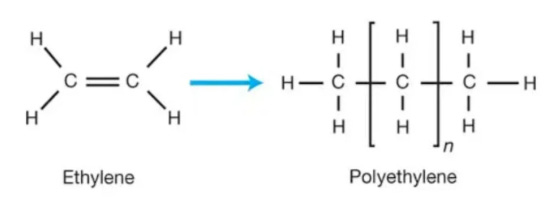
The second (weak) bond is what makes olefins so versatile and important as building blocks. When this bond is broken, the carbon atoms need to join something else to stabilize, and therefore chemists can form new molecules.
For example, by “simply” (in super complex chemical terms) breaking the bond between the two carbons, one can create chains of what seems like ethane (simple bond) molecules, in polyethylene. By adding oxygen, ethylene can turn into ethylene oxide which then becomes part of the PET chain. By adding chlorine, ethylene can become dichloroethane, which eventually leads to PVC. The same can be said of propylene, leading to polypropylene, propylene oxide (important for polyurethanes), acrylonitrile (mixed with ammonia, and important for ABS plastics), and so on.